Depression is a serious mental health problem today and the pathological mechanism of depression is complicated. There are multiple hypotheses about the etiology of depression.
One hypothesis is referred to as “cytokine or inflammatory hypothesis of depression”, which proposes that the actions of inflammatory processes, pro-inflammatory cytokines in particular, on the brain may account for the pathogenesis of depressive disorders. Increasing evidence has demonstrated that both chronic stressors and inflammatory immune activation contribute to pathophysiology and behavioral alteration associated with major depression. Studies from the research group led by Wenjuan Lin demonstrated that both chronic stress and inflammatory immune activation increased central pro-inflammatory cytokines mRNA and induced depressive behaviors. The increased hippocampal TNF-α mRNA expression was the common affected factor in both chronic stress induced and inflammatory immune activation induced models of depression. These results suggest that hippocampal inflammation may have a pivotal role in depression. However, how neuronal-immune interactions modulate the depressive phenotype is not fully understood. Inflammatory processes may affect more than one pathway, which is thought to be important in the pathogenesis of depression, such as monoamine alterations, glutamate neurotransmission, and glucocorticoid receptor resistance. A novel theory suggests that adult hippocampal neurogenesis, as a novel pathway, may also be involved in depressive disorders caused by inflammation.
Neurogenesis is the complex process of generating new neurons from neural stem or progenitor cells that occurs in the brains of many animal species, including humans. Although it was commonly held that neurons in the central nervous system of higher vertebratesare formed during embryonic development and that neurogenesis would not occur postnatally, recent research demonstrated that adult neurogenesis occurs in the subventricular zone of the lateral ventricles and in the subgranular zone of the dentate gyrus in the hippocampus.Recent evidence indicates that neurogenesis is influenced by many factors including inflammatory processes. However, it is unknown how hippocampal neurogenesis is affected by neuro- inflammation and whether the altered hippocampal neurogenesis contributes to the regulation of depression induced by inflammation.
To understand whether and how hippocampal neurogenesis is linked to depression induced by inflammation, an animal model of depression induced by triple central lipopolysaccharide (LPS) administration causing neuro-inflammationwas used by Tang et al. of Lin ‘s research group.The neuro-inflammatory state in the hippocampus evoked by LPS was marked by an increased production of pro-inflammatory cytokines, including interleukin-1β, interleukin-6, and tumour necrosis factor-α. The rats in the neuro-inflammatory state exhibited depressive-like behaviours, including decreased saccharin preference, reduced locomotor activity, and increased immobility time in the tail suspension test and latency to feed in the novelty suppressed feeding test. Adult hippocampal neurogenesis was concomitantly inhibited, which included decreased cell proliferation and a reduction in immature neuron survival. Results demonstrated that the depressive-like behaviours were positively correlated with the inhibition of neurogenesis.The activation of extracellular signal-regulated kinase (ERK), the downstream molecule of fibroblast growth factor 2 (FGF2), was also inhibited by neuro-inflammation. By manipulating adult hippocampal neurogenesis through infusion of FGF2, one crucial molecule modulating cell proliferation and survival in central nervous system, it was found that exogenous FGF2 prevented the decrease in phosphorylation of ERK1/2, reversed depressive-like behavior and alleviated inhibited neurogenesis induced by neuro-inflammation.
These findings (Fig 1) provide evidence that hippocampal neurogenesis dysfunction is linked to neuro-inflammation-induced depression and neurogenesis pathway is a viable therapeutic target to inflammation-associated depression.
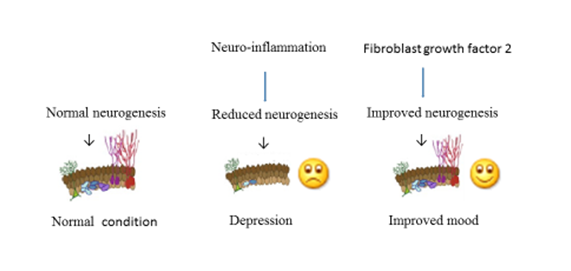
Fig 1. Neuro-inflammation induced impairment of neurogenesis and depression. Fibroblast growth factor 2 promoted neurogenesis under neuro-inflammatory state and improved mood.
Studies were supported by the grants from the National Natural Science Foundation of China (31170987 and 31440045) and the CAS Key Laboratory of Mental Health, Institute of Psychology (KLMH20142G01).
The paper is now available online fromBrain, Behavior and Immunity. Ming-ming Tang,Wen-juan Lin*, Jun-tao Zhang, Ya-wei Zhao, Ying-cong Li(2017).Exogenous FGF2, reverses depressive-like behaviors and restores the suppressed FGF2-ERK1/2 signaling and the impaired hippocampal neurogenesis induced by neuroinflammation. Brain, Behavior and Immunity,http://dx.doi.org/10.1016/j.bbi.2017.05.013
Related paper: Ming-ming Tang,Wen-juan Lin*,Yu-qin Pan,Xi-ting Guan, Ying-cong Li, (2016). Hippocampal neurogenesis dysfunction linked to depressive-like behaviors in a neuroinflammation induced model of depression. Physiol. Behav. 161, 166–173.
Contact:
Wenjuan Lin
Institute of Psychology, Chinese Academy of Sciences
linwj@psych.ac.cn
